There is a quiet shift happening in how scientists think about body fat. Not long ago, fat tissue was seen mostly as storage. Passive. Harmless unless it becomes excessive.
Now the conversation feels different. More layered. Sometimes even unsettling.
Because fat is not just storage. It behaves like an organ. It sends signals. It influences hormones. It shapes inflammation. And when its function goes wrong, something deeper than weight gain may begin.
This condition is called adiposopathy.
What is adiposopathy?
So, what is adiposopathy in clear, everyday language?
It refers to unhealthy or dysfunctional fat tissue. Not simply too much fat. Not just visible obesity. Rather, fat that stops working properly.
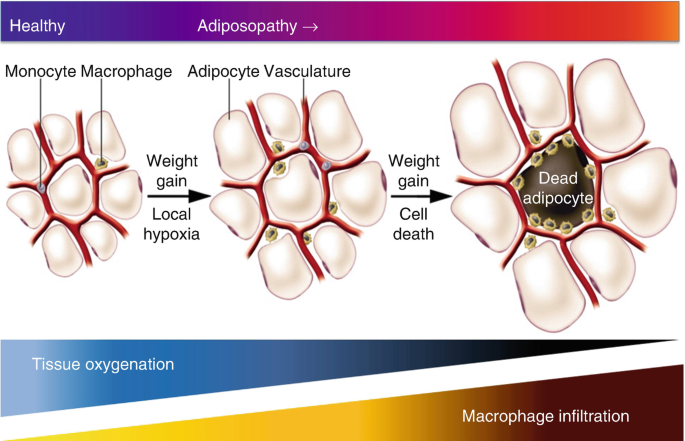
Healthy adipose tissue expands smoothly when energy intake increases. It stores fatty acids safely. Releases them gradually when needed. Maintains metabolic stability.
But under chronic stress from overeating, inactivity, sleep disruption, or genetic vulnerability, fat cells may enlarge excessively. Blood supply becomes inadequate. Inflammatory signals increase. Hormonal communication changes.
At that point, fat tissue shifts from protective to harmful. That shift is adiposopathy.
Some researchers even call it “sick fat.” Slightly dramatic, perhaps. Yet the idea captures the functional disturbance well.
Fat as an endocrine organ
It helps to understand that adipose tissue is not silent. It produces adipokines such as leptin, adiponectin, resistin, and inflammatory cytokines. These molecules influence appetite, insulin sensitivity, vascular health, and immune activity.
When fat tissue functions normally, these signals remain balanced. Appetite regulation feels stable. Energy levels fluctuate naturally.
In adiposopathy, signaling becomes distorted. Leptin resistance may develop. Adiponectin levels often decline. Pro-inflammatory mediators increase.
This creates a metabolic environment where insulin resistance, vascular dysfunction, and systemic inflammation can slowly emerge.
Adiposopathy mechanism
The adiposopathy mechanism unfolds through several interconnected processes.
First, chronic energy surplus drives adipocyte hypertrophy. Fat cells enlarge rather than multiply efficiently. Enlarged cells struggle to maintain an oxygen supply. Local hypoxia develops.
Second, immune cells infiltrate adipose tissue. Macrophages release inflammatory cytokines. Fibrosis may form. Tissue flexibility declines.
Third, lipid overflow begins. Fatty acids escape into circulation and accumulate in organs such as the liver and muscle. This contributes to insulin resistance and metabolic imbalance.
Fourth, hormonal signaling disruptions affect appetite control and energy expenditure. The body may paradoxically increase hunger while reducing metabolic efficiency.
Taken together, these changes define the pathological progression of adiposopathy.
Visceral fat danger
Not all fat behaves equally. Subcutaneous fat under the skin often acts as safer storage. Visceral fat around internal organs tends to be more metabolically active and inflammatory.
Excess visceral accumulation strongly correlates with cardiometabolic risk. Waist circumference becomes a meaningful indicator.
A person might appear moderately overweight but metabolically stable if fat distribution remains favorable. Another individual with similar weight but higher visceral fat may face greater metabolic challenges.
Mayo Clinic confirms waist circumference >35 inches signals visceral fat risk beyond BMI
This variability highlights why adiposopathy cannot be answered by body mass index alone.
Inflammation connection
Chronic low-grade inflammation sits at the heart of dysfunctional fat tissue. Enlarged adipocytes release chemotactic signals attracting immune cells.
These immune cells produce tumor necrosis factor alpha and interleukins that interfere with insulin signaling pathways. Blood vessel function may deteriorate. Lipid metabolism shifts unfavorably.
Inflammation in this context is rarely dramatic. No fever. No acute pain. Instead, there is persistent metabolic friction. The kind that quietly reshapes disease risk over years.
Real-life patterns
Imagine someone working night shifts for several years. Sleep cycles remain inconsistent. Meals are irregular. Stress levels stay elevated. Physical activity declines.
Gradually, abdominal fat increases. Blood pressure rises slightly. Fasting glucose edges upward. Lipid profile worsens.
These changes may reflect developing adiposopathy rather than simple weight gain. Fat tissue itself is becoming less functional.
Another scenario involves childhood obesity followed by rapid adult weight loss attempts. Repeated cycles of gain and loss can alter adipose tissue architecture. Storage efficiency declines. Hormonal signaling becomes erratic.
Adiposopathy and metabolic syndrome
The relationship between adiposopathy and metabolic syndrome is particularly significant. Metabolic syndrome includes abdominal obesity, insulin resistance, hypertension, dyslipidemia, and increased cardiovascular risk.
Dysfunctional fat tissue contributes to each of these components. Inflammatory adipokines impair insulin action. Free fatty acid flux increases hepatic glucose production. Endothelial function deteriorates.
In this sense, adiposopathy acts as a central driver rather than a secondary outcome. Some experts argue it should be viewed as a root pathology underlying modern cardiometabolic disease patterns.
Hormonal disturbances
Leptin resistance represents a hallmark feature of unhealthy adipose function. Normally, leptin signals satiety to the brain. When resistance develops, appetite regulation weakens.
Cortisol dynamics may also shift. Chronic stress promotes visceral fat deposition and worsens insulin resistance.
Sex hormones influence fat distribution as well. Estrogen decline during menopause often leads to central adiposity changes. These hormonal contexts shape how adiposopathy evolves across life stages.
Cardiovascular implications
Dysfunctional fat tissue affects vascular health through multiple pathways. Increased inflammatory mediators contribute to endothelial dysfunction. Lipid abnormalities accelerate atherosclerotic plaque formation.
Over time, the risk of coronary artery disease and stroke may rise. These outcomes rarely feel connected to fat quality at first glance. Yet metabolic research increasingly supports this link.
Reversibility and adaptation
Encouragingly, adipose tissue shows adaptive potential. Gradual weight reduction can decrease adipocyte size and inflammation. Regular aerobic and resistance exercise improves insulin sensitivity and adipokine balance.
Dietary improvements such as higher fiber intake and reduced ultra-processed foods support metabolic recovery. Adequate sleep restores hormonal regulation.
Still, prolonged severe adiposopathy may leave residual structural changes like fibrosis. This highlights the importance of early metabolic awareness rather than reactive treatment alone.
Emerging research
Scientists are investigating therapies targeting adipose inflammation pathways, mitochondrial function, and adipokine signaling. Brown fat activation strategies aim to enhance energy expenditure.
Microbiome modulation is another frontier. Certain gut bacteria profiles appear linked to improved fat tissue metabolism.
Pharmacological agents addressing insulin resistance and appetite regulation may indirectly improve adipose function. However, lifestyle foundations remain central in most evidence-based approaches.
Johns Hopkins studies confirm microbiome influences adipose inflammation
Practical awareness
Detecting dysfunctional fat tissue is not always straightforward. Indicators may include central obesity, elevated triglycerides, reduced HDL cholesterol, increased fasting insulin, and persistent fatigue.
Yet numbers alone cannot capture the full picture. Movement patterns, stress exposure, dietary quality, and sleep rhythms influence adipose biology profoundly.
Developing curiosity about these factors may help individuals recognize early metabolic shifts associated with adiposopathy.
FAQs
Is adiposopathy the same as obesity?
Not exactly. Obesity describes excess body fat. Adiposopathy refers to dysfunctional fat quality and signaling, which can occur even in individuals without severe visible obesity.
Can exercise improve adiposopathy?
Regular physical activity enhances insulin sensitivity, reduces inflammation, and improves adipokine balance. Over time, this may restore healthier fat tissue function and metabolic stability.
Does diet influence adipose tissue health?
Yes. Diets rich in fiber, unsaturated fats, and minimally processed foods may support better adipose metabolism, while chronic excess calories and refined sugars worsen dysfunction.
Why is visceral fat more harmful?
Visceral fat releases more inflammatory cytokines and free fatty acids into circulation. This directly impacts liver metabolism, insulin sensitivity, and cardiovascular risk profiles.
How fast does adiposopathy develop?
Usually gradually occurs over the years of metabolic imbalance. Rapid onset is uncommon unless hormonal disorders, genetic conditions, or severe lifestyle disruptions accelerate adipose dysfunction.
Can weight loss reverse adiposopathy completely?
Early stages often improve significantly. However, long-standing inflammation and fibrosis within fat tissue may not fully normalize despite substantial lifestyle interventions.
Are blood tests enough for diagnosis?
They provide useful metabolic clues but cannot directly measure fat tissue function. Imaging studies, waist measurements, and clinical context help build a clearer assessment.
Is adiposopathy linked to diabetes risk?
Yes. Dysfunctional adipose signaling contributes to insulin resistance, altered glucose metabolism, and increased likelihood of developing type two diabetes over time.
A deeper metabolic perspective
Perhaps the most interesting aspect of adiposopathy is how it changes our view of fat itself. Instead of focusing only on weight or appearance, attention shifts toward tissue health and metabolic communication.
This perspective invites more nuanced conversations. Not blame. Not simplistic calorie equations. But recognition that modern environments shape biological responses in complex ways.
Understanding what adiposopathy is may encourage earlier lifestyle adjustments, better clinical screening, and more compassionate public health strategies.
After all, metabolic health is rarely determined by a single choice or moment. It evolves through patterns repeated quietly across years. Observing those patterns with curiosity might be one of the most powerful preventive tools available.